I’ve been neglecting to write this blog for the last few weeks, what with the usual Christmas rush. Now that New Years is done, I’m resolved to write more often, at least twice per week. Another reason I’ve been neglectful is that I’ve been quite busy working on episodes of the videos for The Elements Unearthed project, especially the episodes on the history of the periodic table where I interviewed Dr. Eric Scerri of UCLA. He is the author of The Periodic Table: Its Story and Its Significance by Oxford Press.
During the last few weeks I’ve transcribed his interview and sent it to him to look over for revisions, as well as the drafts of the episode scripts. He has been most gracious to provide suggestions that have greatly improved the scripts. Because of the detail of the interview, I’m going to divide it into two parts, the first on the precursors to Mendeleev and the second on Mendeleev and beyond. Each should be about 15 minutes when complete. I will upload a compressed version of each episode here once they are done (another two weeks, tops – I have quite a few client projects happening right now, too) as well as the finished transcript of the interview and the episode scripts. I’ll also upload them to a dedicated video site and then uplink them to iTunes and YouTube.
In preparation for these episodes, I’ve been cleaning up the photos I took this last summer at the Chemical Heritage Foundation of the Edward G. Mazurs collected notes, which he prepared over several decades for his book Graphical Representations of the Periodic System During 100 Years, which he self-published in 1957 and which was then revised and published by the University of Alabama Press in 1974.
He classified over 700 different periodic tables, and his notes filled ten three-ring binders. I also was able to photograph the production artwork that was used for the books. It dawned on me while I was doing the clean-up that I didn’t actually have any photographs of the final books, so I traveled over to Brigham Young University’s library two weeks ago and found both editions on the shelves, as well as Jan van Spronsen’s book and a book in Russian with photos of Mendeleev, his notes. and his laboratory. I photographed all the relevant pages, including any photographs or portraits of the people who contributed to the development of the periodic table, including such people as Alexandre Emile Beguyer de Chancourtois, who developed his Telluric Screw in 1862 which shows the first discovery of the periodic law: that the properties of the elements seem to repeat periodically.
Finding the 1957 edition of Mazurs’ book is quite rare, since not many were printed. While I was there, I looked up an article I remember reading in Chemistry magazine back in the 1970s on various forms of the periodic table. It’s funny how memory can play tricks on you, however. What I thought was a major article showing various forms of the table in full color was actually a short article showing one form of the table (although it was in full color). I apparently have a better memory for images than for text; my memory had expanded and aggrandized the article into something much more than it was. But the table was interesting, and here is a photo of it:
I was also struck as I was preparing these images from Mazurs’ notes how some of the more exotic continuous-form periodic tables look remarkably like images of strange attractors in fractal mathematics. I’ve been playing around with an interesting free-ware program called Chaoscope trying to come up with similar images and here are a few samples comparing Mazurs’ notes and artwork with fractal patterns. Wouldn’t it be fun if some bored mathematician was able to show that the unusual pattern of the periodic system (created by the quantum mechanics of electron orbital filling in successive atoms) followed a fractal equation? I’m afraid I’m not much of a mathematician, but I can make some pretty pictures now and then. Anyway, from a visual standpoint, the similarities are amazing.









I thoroughly enjoyed your article. Phil Stewart of Oxford will
like it, as will Ted Benfey who created the “snail” table.
Mazurs book should be used with great care. He redrew the images, systematically adding elements that were not known when they were first published – which is acceptable – and changing their shape – which is totally unacceptable (for example figure 111 which hideously misrepresents Janet’s elegant original). Sometimes he misattributes them, for example he credits Janet with a design that is not his at all (fig. 85, which is a distorted version of Stedman’s). If he doesn’t like something he simply leaves it out, for example the element zero in tables by von Antropoff and Janet. I do not trust any illustration of Mazurs; you should always check against the original. This is unlike Van Spronsen who is scrupulously accurate.
Mazurs book suffers also from the fact that he classifies the images in a system that is almost incomprehensible, and his bibliography and index are not cross referenced to page numbers, so that it is a heroic enterprise to find anything.
One thing that I approve in both Mazurs and van Spronsen is that their titles refer to the Periodic System. Tables are just one way of representing the system, which is ultimately an abstract mathematical object. Its best two-dimensional representation is a spiral, which does not break up the continuous sequence of elements. A ribbon wound in three dimensions is even better (first adopted by Courtines in 1925).
Thank you for your comment – Yes, I’ve noticed some of the inaccuracies in Mazurs’ illustrations, although I haven’t looked at many of his sources yet. It’s interesting to see his original notes and compare it with the artwork produced for the books. The artwork appears to have been created based on his notes, because he has the figure numbers written in red pencil by his drawings and appears to have merely given his notes to the artist, not the original articles. He largely hand drew copies of the original illustrations with only a few photocopies included. In many places he’s gone back and redrawn the same table and pasted it over his original drawing using rubber cement (which has created dark brown spots because of the acidity of the paper). So it’s not surprising that errors and distortions crept in. It’s good to know that Van Spronsen was more careful. I also agree that Mazurs’ system of classification is unusable. At least his 1974 edition has the artwork integrated with the text, whereas the 1957 edition had all the tables at the back of the book and no index to speak of.
Do you have a reference for the Courtines 3D ribbon system? I’d like to try to recreate some of these 3D systems in a 3D modeling program as part of the video I’m putting together.
You mention de Chancourtois, developer of the first true periodic table, his Telluric Screw, but treat that as if it were a precursor to graphic table rather than a spiral 3D model with all sequential elements adjacent and still vertically aligned in groups.
The Alexander Arrangement of Elements is a successor the Telluric Screw in that the plane of the element data portrayal is curved, including the elements de Chancourtois had to work with and all the others identified to date – with the Actinoids and Lanthinoids integrated.
Thank you for the comment on de Chancourtois. Yes, I should have mentioned that not only was his attempt the first true periodic system but it was also the first three dimensional system or the first continuous form table if we try to show it in 2D. For the video project I’m working on, I will re-create his Telluric Screw in 3D and show the alignment of the elements more accurately through a 3D anmation.
Do you have a reference for the Alexander Arrangement that you mentioned? I’d like to see any of the modern attempts to update some of these pioneering periodic systems.
Courtines’ article ‘A model of Periodic Table’ is in J. Chem. Educ. 2, pp. 107-09 (1925). Incidentally Mazurs bibliographical references are scrupulously accurate – when you can find them! Van Spronsen has a picture of it, p. 183. Courtines rather spoilt it by cutting the ribbon at the end of each period and indicating the join by heavy black lines.
Roy Alexander’s arrangement is basically that of Courtines, joined up properly, with the lanthanoids (and actinoids) expanded. Rather surprisingly he was able to patent the idea, but you can easily make your own ribbon.
Another variant of the three dimensional spiral was that of D F Stedman; ‘A Periodic Arrangement of the Elements’, Canadian J. of Research, 25 B, 199-210 (1947), images in van Spronsen, pp. 185-6. Its 2D projection is basically the same design as was found independently by Ted Benfey (described in a recent article in J. History of Chem).
You can’t make a simple spiral because the length of the coils increases only after every second one. In my ‘Chemical Galaxy’ I tried to get round this by making their length increase regularly and using curved radial lines to join up the groups.
Thank you for asking, David; the Alexander Arrangement and its rationale can be found at http://AllPeriodicTables.com.
While the advanced study and profession of Chemistry has no need of more dimensions than two for arranging element data for reference – convenience and multiplicity of applications demand flat formats – the introductory hurdle of the very new chemistry student, that dreaded icon of educational difficulty, the STANDARD PERIODIC TABLE, needs to be cleared first.
Philip Stewart, myself, and others have attempted to help in this process, introducing the missing continuity and contiguity in different formats; his through dramatically picturing dimension and drawing connecting lines, and mine by physically attaching all the related data boxes on a 3-dimensional object.
who is 3dpt? or must this remain incognito?
Hello Mike,
3dpt is me, Roy Alexander…and I’m feeling good today!
Yesterday I discovered that de Beguyer, Courtine, Denker, and I are joined by George Gamow, a VERY respected world famous scientist, in our independent development of similar 3D periodic tables.
Eric Scerri called my attention to part of an article at http://www.informit.com/articles/article.aspx?p=423773&seqNum=3 .
The article is apparently part of a book by scientist/author George Gamow (prounounced GAMM-OFF), and, it seems, the developer of a virtually identical arrangement of the elements to the Alexander Arrangement, and technical reasons described by him.
His drawings are at the bottom of that page, and with mine at http://www.allperiodictables.com/3d/3dPTpatentdrawings.jpg
The drawing technique used in his illustration is (or was in the 60s at least) precisely the standard style required for submission to the patent office, so it appears that he was moving in the same direction as I. Perhaps bad health intervened, as he died in 1968.
While the year of my invention was 1965, in Chicago, my patent was issued in 1971. I had started work on the application drawings perhaps a year or so earlier – back East, nowhere near Boulder, CO, where George Gamow lived & worked.
I am not the only one to be “amazed to see how little interest there is in 3D periodic tables in the chemistry-teacher community” a recent communication states, “You’d think every chemistry teacher would want one for his [or her] desk, if not one for every student.”
Roy
Roy:
Thanks for the comments! I’m in the process of trying to put some of these periodic tables, both 2D and 3D, into a series of animations to add visual interest to the podcast episodes I’m editing. I’ve completed what I think is a decent 3D model and animations of de Chancourtois’ Telluric Screw, and now I’m building a series of 3D element tiles that will have modern atomic weights (rounded to the tenth decimal place) and are color coded by families (such as the alkaline earths). What I intend to do is take the tiles and animate them moving into position to form a series of periodic tables. Some of the shapes employed in 3D tables (such as your ribbon shape) are too complex to easily model and texture in 3D space, but using a series of tiles that can be stacked and shuffled around will resolve that problem. I will put the tiles close together so that there will not be gaps and give the animations a continuous appearance. With your permission, I would like to create a version of your table using these tiles and show it in the video episode, giving you proper credit, of course. I’ll send you copies of the images and animations rendered from the model that you can use however you’d like. I’m still shooting for final upload of the episodes by the end of next week, and am working on what we call the “B-roll” materials, such as animations, images, titles, and such. I’ve got all the primary “A-roll” footage ready to edit, including the narration. I will be adding another blog entry tomorrow to follow up on this “Strange Attractor” post that has generated all these comments, and I plan on including the basic script of both episodes so that all of you can look if over and make sure I”m not leaving anything out (keeping in mind that my audience is first year inorganic chemistry students).
Dave Black
Previous post’s boldface is accidental.
Roy
There are at least two “3dpt”s. With google I found an astrologer and homeopath in New Zealand with a web-page called that. He sells his own arrangement on three disks at right angles to each other. I find it strange that anyone has patented a design. The ribbon is the simplest possible way of representing the Periodic System, but it can be wound and coloured in any of a large number of ways, of which Roy’s is only one. In fact periodic tables are just ways of cutting and pasting the ribbon, and periodic spirals are the ribbon wound in two or three dimensions.
Dave:
I am very happy to have the AAE used as you describe, and look forward to incorporating whatever animation you arrive at on my soon-to-be revised website!
Some time ago I tested a potential product, which employed the equivalent, I believe, of the tiles you mention.
There are images online which may be of help to your people; of the descriptive flier, at http://allperiodictables.com/2009_AABB/.
The down slant in the P-block (connecting the periods without loss of contiguity of the groups), which is the core of the Alexander Arrangement of Elements, can be handled as shown on the flier, with multiple steps until the level of the next period is reached.
Page 2 of the flier shows the attachment of the F-block differently than ordinarily seen, but one of the features of the AABB is that students may try any arrangement – even a flat table for a big empty wall. The latter releases the set-up area for other uses, and acts as a gallery for the creative work various students have put in on the individual element ‘tiles’.
Roy Alexander
Hi Philip,
About the 3dpt: When signing up for the blog I chose a nom de blog before recognizing that others were using their full names. Sorry to create a mystery. I am not hiding. I will try to enter Roy Alexander in the Name box when I post this.
About a patent: The work I did developing my 3d periodic table was original, as far as I had found with limited research. Assuming that the scientific community would recognize its value, I planned to invest in production of them for school use (and did so, in several versions).
A patent is to protect individual inventors like me from larger firms pinching a creative idea and producing and marketing it faster, better, cheaper, and leaving us in the dust.
When what the patent office calls Prior Art is discovered by an inventor or the patent examiner there can be no patent issued, nor is it necessary, as with the Longman type spiral or the standard flat periodic table.
About the Element Ribbon;
It is, as you say, the simplest possible way of representing the Periodic System, and why the most daunting and confused way, the standard table, with the ribbon cut a dozen times or so, the icon of educational difficulty, is used for beginning students is beyond comprehension.
For the ribbon to become the best way, however, without losing the simplicity, it must be wrapped so the groups with similar properties can be brought together.
The Longman and similar spiral illustrations (http://allperiodictables.com/aptpages/gridlink/grid07_spiral.htm ) indicate desired group and element connections with lines or a wide variety of box sizes on a poster, so they are not easily lumped together with the Alexander Arrangement.
I have described my steps to the invention as starting with such a ribbon, but have ended with a planar helical model with same size element data boxes (except for special treatment of H, whose contiguosity, so to speak, seems much in demand) capable of bearing as much element data as desired, with every element box contiguous with both the next and previous elements in both periods and groups, from period to period, and with the blocks properly connected as well.
The AAE is not intended to be a new system; it does not add, subtract, or duplicate elements, but provides the new student with an uncomplicated way to start learning about elements, by seeing them arranged intelligently and simply in a widely acceptable way.
If scientists can find new and interesting relationships by looking around corners, then all to the better. I hoped that would be the case.
Roy Alexander
David,
If, in your video presentation, aspects of the AAE are to be mentioned, perhaps the following would be interesting points to be considered;
1. the table conforms strictly to Mendeleev’s line – without cuts, and following the n+1 (Madelung) rule. This includes no cuts at the ends of periods or blocks as well, and no spaces (as in spiral illustrations) anywhere.
2. The (single) H box (alone) is elongated, which permits H to be touching on Be, Li, F, and He, either with a full or part of a side or a corner. The rationale for these connectivities(?) or kinships, whether they be primary, secondary, or tertiary, will have to be determined by others. Other relationships with element groups have been championed by one or the other of periodic table afficianadoes, and the long strip, which is the H box, soars over all the element groups of the s- and p-blocks, which might satisfy a few.
3. Some primary, secondary, or tertiary kinships have been identified as occurring with the AAE that can not be found on a flat chart. Except for H, above, primary kinships shown are pretty much the same as on most flat tables. The extra dimension permits an element data box corner on one plane to be in contact with a corner on another plane.
Therefore; secondary kinships shown corner to corner on the AAE are Al-Sc, and Y-La, and not on flat tables, and tertiary kinships shown only on the AAE, also corner to corner, are of Mg-Zn and Sr-Yb.
Have I sent you an AAE model so you can see for yourself?
Roy
Hello David,
Any news about the computer animation of the AAE?
Can I help?
Roy
Roy:
I got looking at how I would try to create a 3D model out of the tile objects I’d used for the other animations, and I couldn’t see how to do your Alexander Arrangement justice – it wouldn’t have had the right curvature and look, especially in the first few elements. I did find a photo online of Glen Seaborg holding one of your completed Arrangements, but it was too low resolution to use in the final video. Did you present him with that?
David Black
David:
I’m surprised at the difficulty. Nothing could be simpler.
I just put online an article with illustrations of how 5th graders can make a 3D periodic table out of a standard flat table. (see http://www.allperiodictables.com/LeftStepAAEOnline/ )
I started with a left-step table (also online), but starting with the standard long form table works just as well, as all the gaps are closed ultimately in any case. Once the first step, make a circle, is done, they are alike. (Using the standard short form means inserting the Rare Earths first.)
I left off the H and He because of the contentiousness on the Sciencebase Blog, but that is a simple wrap as well, H starts overLi, and extends around to join with He, which is over Ne. (see http://allperiodictables.com/kin )
I did send you a DeskTopper, didn’t I?
I have put a Seaborg hires photo at http://allperiodictables.com/3d/ap.wwp.Seaborg.jpg for you. The photo with his “favorite periodic table” (according to the photographer, Susan Ragan) was used in the Guinness Book and world wide for most of his obituaries. The Rare Earths, which he had displaced from the main body of the table is the loop in his right hand on his model which I had made for him – well in advance of the official naming of 108 – near his left hand. (Please attribute AP World Wide Photo.)
Roy:
The difficulty I’ve had wasn’t with making a physical paper model, but with creating a virtual 3D model with animation (similar to what you see in the videos with the tiles shifting to become a left-step table, for example). I can’t get the solid blocks to wrap around naturally enough.
Thanks for the higher resolution photo of Glen Seaborg. I will be creating more videos on the periodic table (properties of the elements, etc.). I don’t think you’ve sent me a paper model.
David Black
I can send one if you provide an address (preferably by email).
Roy@allperiodictables.com
Roy:
My e-mail address is: elementsunearthed@gmail.com
Thanks!
David Black
It seems that working with 3D blocks to picture a tabular item is the hard way, and I can see that determining what hinges this way or that for a looping and folded sheet could be problematical, even with the vertical plane always the same.
Roy
The videos are great!
Just right for the YouTube format. Love the graphics and the emphasis on de Chancourtois.
Sorry that the AAE, successor to the Telluric Screw couldn’t get in there.
Good Luck with them, and keep up the good work.
Roy
Dear Mr. David Black
Very interesting your work of rescuing the contributions about of the PT., especially of Mazurs, and the genial work of C. Janet. It really is invaluable that all this can be seen around the world, previously impossible, the magic of the Internet brings us closer to understanding the science with such precision and astounding beauty.
For my part I made a development of the Chancourtois telluric screw, and I have a mathematical studies on mathematical functions or matrices that make up the P.T.
I am currently finalizing a new version of my studies, in which I have found a mathematical relationship that I believe it define a new quantic number, and is the concept of Binode (a pair of atomic levels or a pair of periodic in the Left Step Janet P T, in the Dr. Scerri’s book, page 283 ) so I’ve designed a new table called “Binodic Table”, and that corresponds to a simple mathematical expression: a parabolic curved
Addresses or links are as follows. The paper it is in Spanish language.
http://www.monografias.com/trabajos-pdf/tabla-periodica-nuevo-modelo/tabla-periodica-nuevo-modelo.shtml
DNA of the Materia or the telluric screw of Gutierrez Samanez
My best wishes to you.
Julio Gutiérrez Samanez
From Cuzco, Peru
There is a much simpler way to make a 3-D model. Mazurs illustrates it: Giguere’s helix (1966). It’s a stylized form of the Schaltenbrand helix. It combines the best features of the spiral and series tables. You split each block in two, and glue them back to back – s and d on one axis, f and p on the other – cut a notch in the last two periods between s and d block, and clip this over the other axis between f and p block. It’s compact, easy to make, and cheap to print (it fits on one A4 sheet). It can carry a wealth of detail – it looks great with the blocks in different colours, and with photographs of the elements. The electron configurations are easy to work out. It’s true to the electrons’ filling order, showing both Hund’s rule and the Pauli exclusion principle. Its only drawback is the Janet problem of helium being separated from the noble gases.
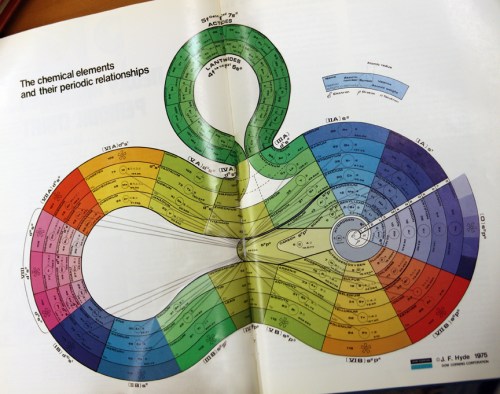
Spronsen shows one spiral which children love – Romanov’s 1934 lemniscate, which is fairly easy to update and colour in Photoshop. It unites the main block elements above the transition elements, and suggests rising energy levels by threading f block through the middle. It’s the best for showing how the initial cut, before the halogens, or after the noble gases or alkaline earths, produces the radically different Scerri, Werner, and Janet tables. Its main drawback is it places lanthanum below scandium, but that just shows how each table has its own agenda, which is a valuable lesson in itself..
Giguere’s beautiful architectural model is one of the few that Mazurs illustrates accurately (and it is missing from van Spronsen). I don’t see why He over Be is “the Janet problem”. If the s block is to be kept separate from the p block it is Janet’s solution, and it was adopted by Simmons, Giguere, Mazurs van Spronsen, Bent, Katz (and Scerri, till he changed his mind and decided to forget about orbitals and cut the ribbon between the oxygen group and the halogens). The s-block/p-block boundary is the only one where there are no embarrassing ambiguities like La, Ac, Th, Zn, Cd, Hg. There is no way He can be called a p-block element. Its noble-gas-like behaviour results from the fact that there are only 2 electrons in the K shell. All the other shells can form an octet with 2 s and 6 p electrons.
Clark in Life magazine, copied by Longman, pioneered the use of colour to show secondary relationships between transition elements and representative elements (Mendeleev’s A and B columns within his groups). I took this over in my Chemical Galaxy II, extending it to the lanthan/actinides to pick up the II valency in Eu and Yb, III in La and Gd, and IV in Ce and Tb, with the rest echoing Mendeleev’s group VIII in having less valence electrons than might be expected.
The picture labeled as the Lorenz attractor is not the Lorenz attractor. Instead it is another, somewhat simpler attractor known as the Rössler attractor.